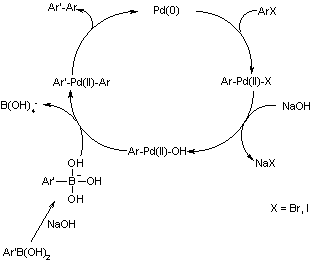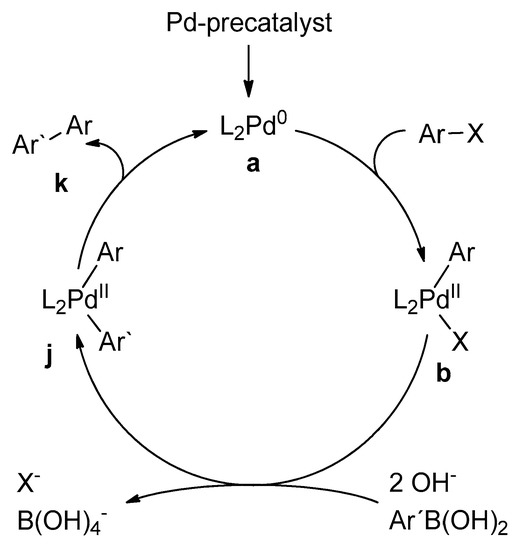
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross- Coupling Reactions: Generation of Carbon–Carbon Bond

Palladium-Catalyzed Suzuki–Miyaura Coupling of Aryl Esters | Journal of the American Chemical Society
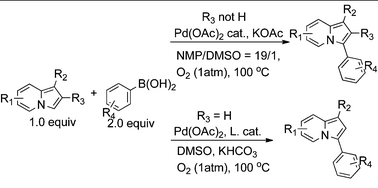
Palladium catalyzed oxidative Suzuki coupling reaction of indolizine at the 3-position using oxygen gas as the only oxidant - RSC Advances (RSC Publishing)

The Synthesis of Biarylmonophosphonates via Palladium-Catalyzed Phosphonation, Iridium-Catalyzed C-H Borylation, Palladium-Catalyzed Suzuki–Miyaura Cross-Coupling | Catalysis Letters
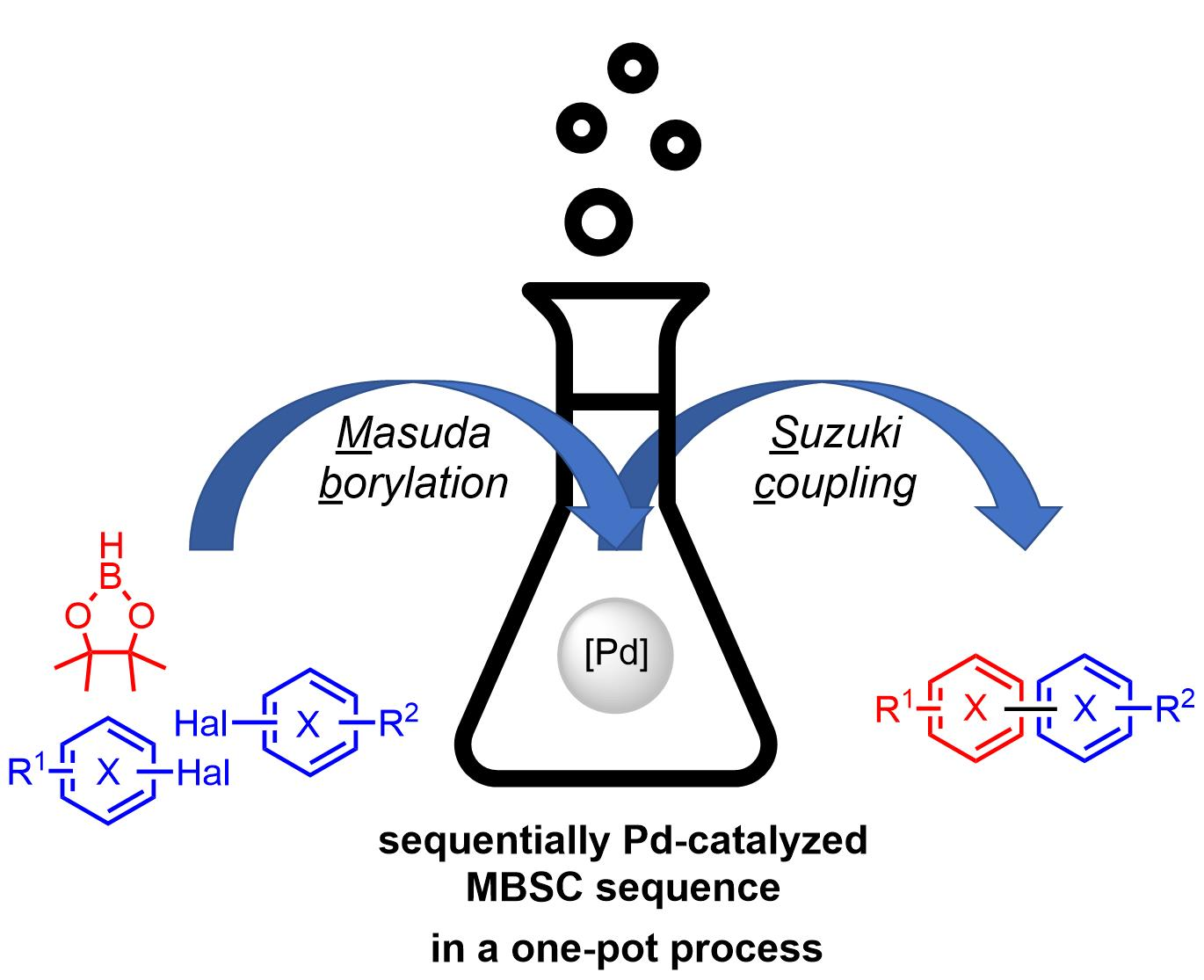
Catalysts | Free Full-Text | Masuda Borylation–Suzuki Coupling (MBSC) Sequence: A One-Pot Process to Access Complex (hetero)Biaryls

Palladium nanoparticles catalyzed Suzuki cross-coupling reactions in ambient conditions - ScienceDirect

Biogenic palladium nanostructures for Suzuki-Miyaura and Sonogashira cross-coupling reaction under mild reaction conditions - ScienceDirect

Ligand-Free Palladium Catalysis of the Suzuki Reaction in Water Using Microwave Heating | Organic Letters

Ligand-free Suzuki coupling reaction with highly recyclable ionic palladium catalyst, Ti1-xPdxO2-x (x = 0.03) - ScienceDirect

Palladium-Catalyzed Suzuki–Miyaura Coupling of Aryl Esters | Journal of the American Chemical Society
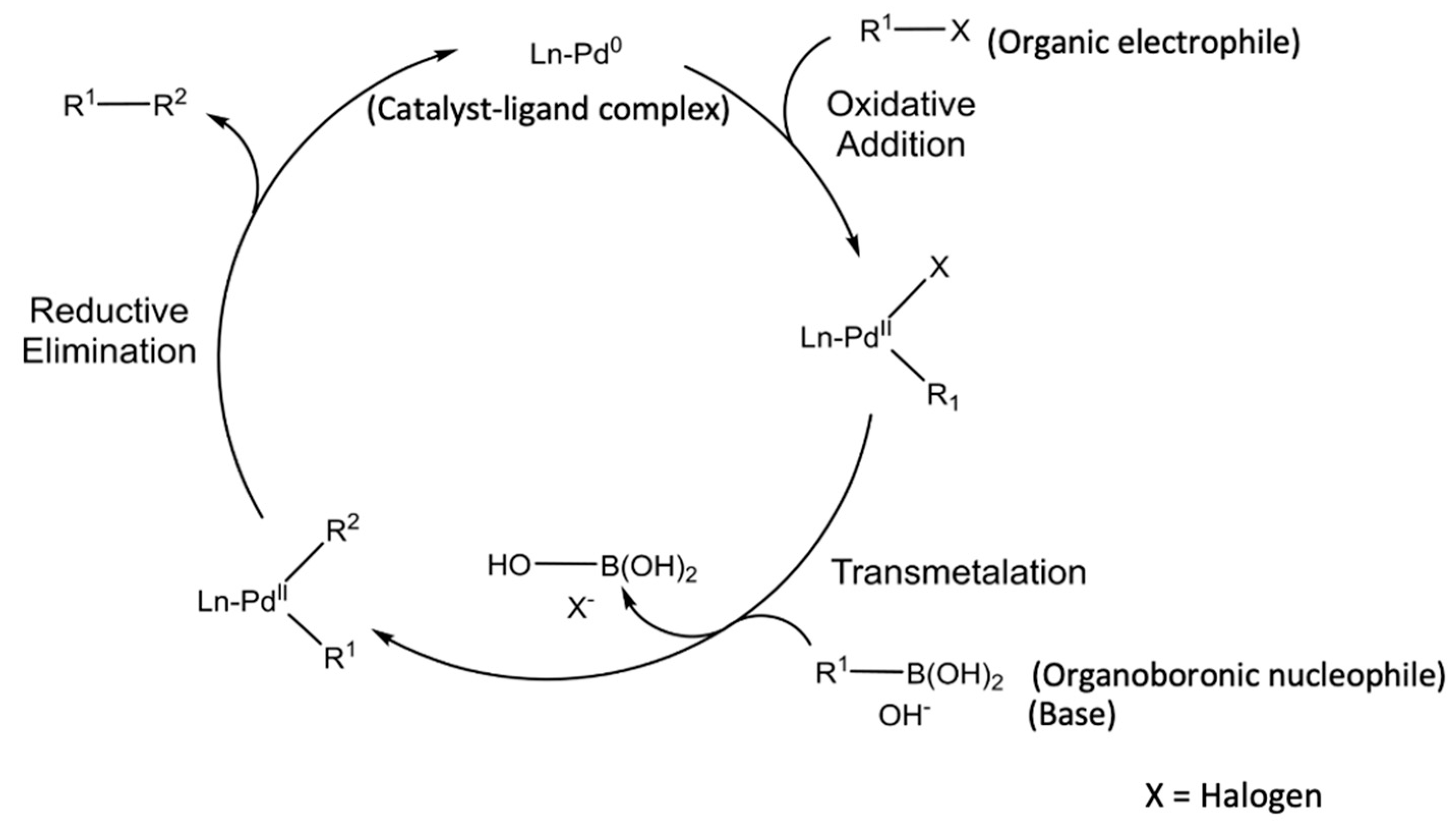
Knowledge | Free Full-Text | Catalyst Recycling in the Suzuki Coupling Reaction: Toward a Greener Synthesis in the Pharmaceutical Industry

Palladium-Catalyzed Suzuki–Miyaura Coupling of Aryl Esters | Journal of the American Chemical Society
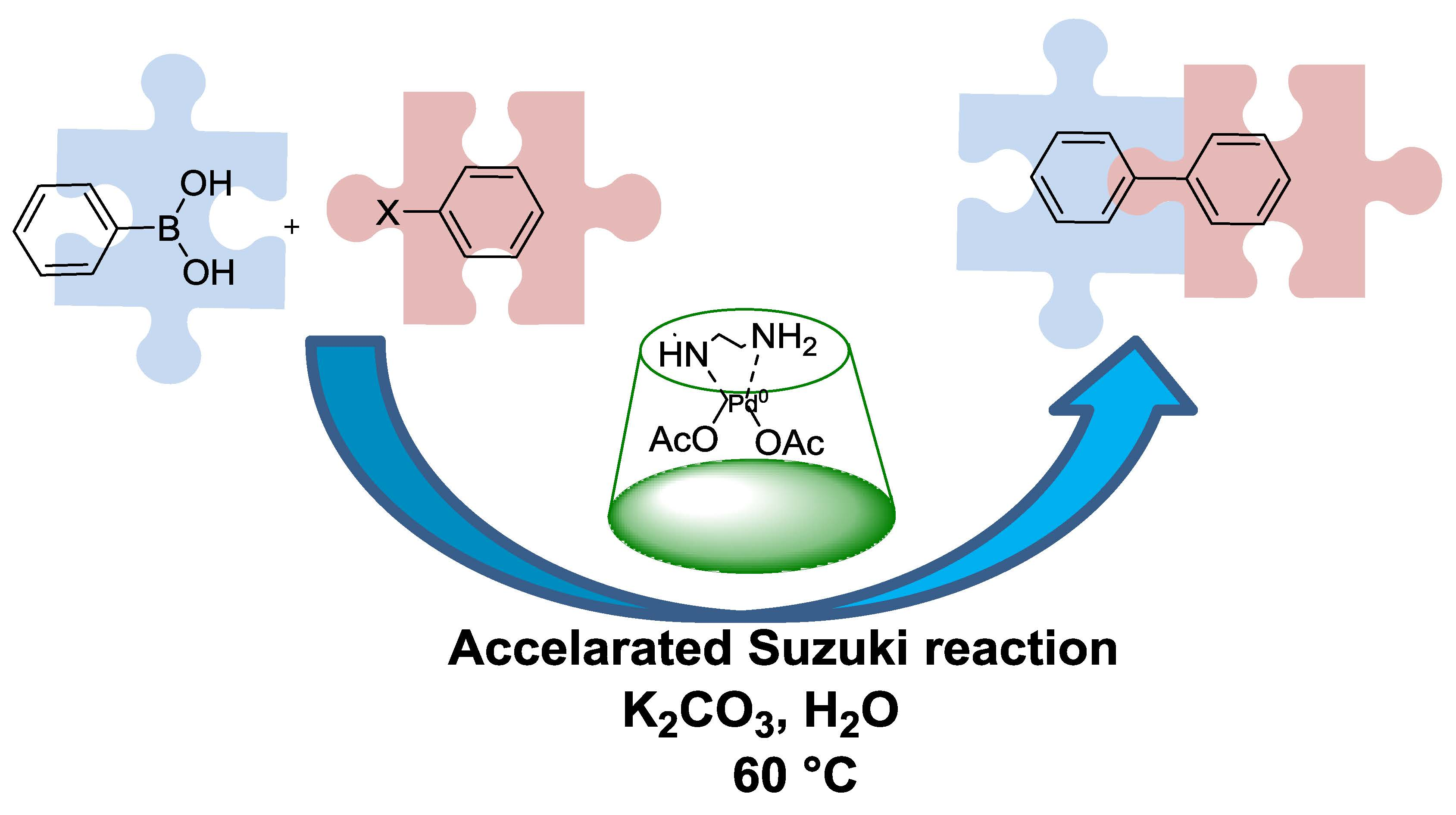
Catalysts | Free Full-Text | An Amino-Chain Modified β-cyclodextrin: A Supramolecular Ligand for Pd(OAc)2 Acceleration in Suzuki–Miyaura Coupling Reactions in Water
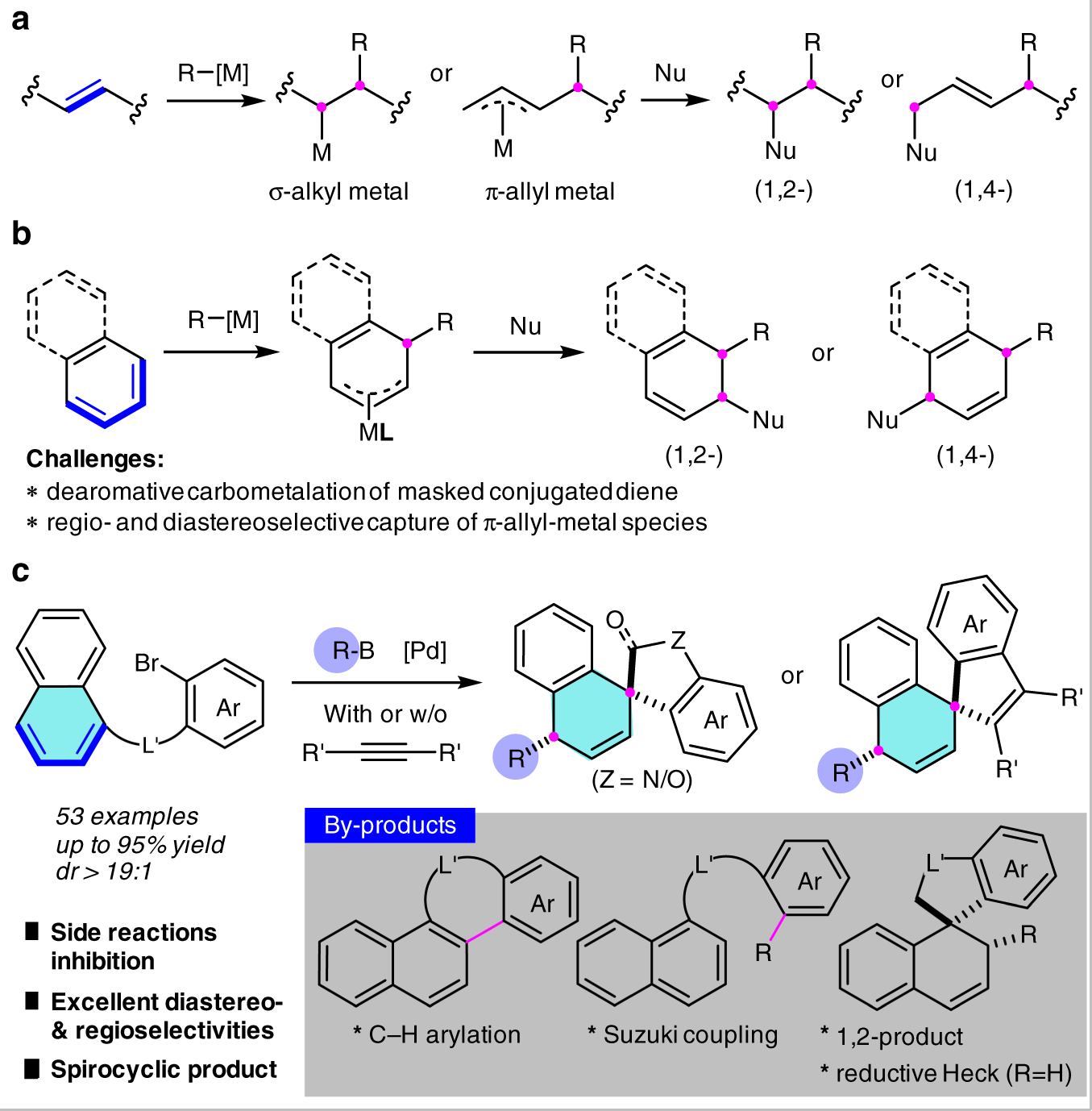
Dearomative 1,4-difunctionalization of naphthalenes via palladium-catalyzed tandem Heck/Suzuki coupling reaction | Nature Communications

Homogeneous and Recyclable Palladium Catalysts: Application in Suzuki–Miyaura Cross-Coupling Reactions | Organometallics

Palladacycle-Catalyzed Triple Suzuki Coupling Strategy for the Synthesis of Anthracene-Based OLED Emitters | ACS Omega

Palladium-catalyzed, ligand-free Suzuki reaction in water using aryl fluorosulfates. | Semantic Scholar

A Combination of Biocompatible Room Temperature Ionic Liquid and Palladium Catalyst for Base‐ and Ligand‐Free Suzuki Coupling Reactions - Joo - 2020 - Asian Journal of Organic Chemistry - Wiley Online Library